
Reduce the impact of noise from shipping on marine species
March 2, 2021
New tests and computer simulation methodologies for electric vehicle chassis weight reduction
March 16, 2021Researchers from research groups at Computational Biology and Complex Systems (BIOCOM-SC) and ' Heart Rate Analysis (ANCORA) from the UPC, along with Others from the University of Calgary (Canada) , from the Barcelona Institute for Biomedical Research (IIBB-CSIC ) and the Research Institute of the Hospital de la Santa Creu y Sant Pau (IIB Sant Pau) , have identified 1 of the molecular mechanisms responsible for cardiac alternation, an alteration of the heart rhythm that facilitates the induction of ventricular fibrillation, a potentially lethal arrhythmia.
Cardiovascular disease is one of the leading causes of death worldwide, accounting for about 18 million deaths each year. In Spain, and in Catalonia, they represent almost a third of deaths, and it is one of the diseases that also causes a significant decrease in quality of life.
Cardiac alternations have been known since the late 19th century, when an alteration in the pulse (hence the name, pulsus alternans ) was described that alternates a strong beat with a weak one. Almost a century later this alteration was related to the propensity to suffer episodes of ventricular fibrillation, in which heart cells suddenly stop beating synchronously, in many cases with fatal results in minutes. . Since then, work has been done to find out the molecular mechanisms responsible for this arrhythmia, which would open the door to the development of new pharmacological treatments.
Computational models to study alternates
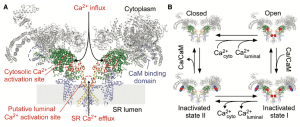
The UPOC BIOCOM-SC group has used computational models to study the origin of cardiac alternations. In most cases, the alternations are due to irregularities in the regulation of dissolved calcium within the cell. Mathematical models predicted that alternates could appear due to alterations in the protein that regulates calcium release in the cell, the rianodine receptor (RYR).
The molecular mechanism responsible for this alteration has been the subject of debate over the last decade where different origins have been proposed for RYR malfunction. The rianodine receptor is a protein found in the inner membranes of the cell where large amounts of calcium are deposited. The RYR behaves like a door that when opened releases stored calcium, activating heart contraction. However, if calcium deposits are overloaded, the door opening is out of control leading to irregularities in the released calcium and cardiac arrhythmias. This study has managed to identify the key molecular mechanism in the regulation of the opening of this door.
Thus, it was studied that this alteration in receptor function could be due to its binding to another protein (calmodulin or CaM), which regulates its function. To confirm this, an experiment was designed to change the function of CaM by introducing into mice adenoviruses modified to produce either the protein in its normal state or mutations in the protein that increased or decreased its function. observing whether this decreased or increased the propensity to develop alternates.
The analysis of these data, using statistical techniques, was carried out by the ANCORA group of the UPC, and concludes that a decrease in CaM expression correlated with a decrease in the propensity to develop alternates.

Experimentally, however, it is not possible to observe what happens with RYR, so there is always the possibility that CaM is affecting another regulatory mechanism, and that there is an alternative explanation for this effect. To confirm this, researchers from the BIOCOM-SC research group have used a computational model that describes in detail the interaction between CaM and RYR and its effect on cardiac dynamics. The results of the mathematical model agree perfectly with what is observed experimentally.
Researchers do not rule out that there may be other factors that affect the appearance of alternates, but the existence of a clear molecular mechanism that leads to the appearance of cardiac alternates may lead to the development of pharmacological treatments for this. arrhythmia
This study on ventricular dysfunctions arises from research on atrial fibrillation and the development of models for the atrium, funded by the Ministry of Science and Innovation, the Generalitat de Catalunya and the Marató TV3 Foundation.
This finding, published in the journal ' Circulation Research' (AHA Jounals) , opens the door to new pharmacological treatments.
Technology
Topic
You want to know more?
Related Projects
- A team from the Bioinspired Oral Biomaterials and Interfaces (BOBI) research group at the Department of Materials Science and Engineering (CEM) of the Universitat Politècnica de Catalunya - BarcelonaTech (UPC) is taking part in the European project HYDROHEAL, which explores how to transform bone fracture treatment using smart and advanced biomaterials, aiming to reduce the risk of infection and implant rejection, as well as shortening fracture recovery times.
- Neurodegenerative diseases, such as Parkinson's disease, Alzheimer's, and age-related disorders, have been widely studied due to their significant impact on individuals and society. So far, these are incurable and debilitating diseases that lead to progressive degeneration and death of nerve cells, resulting in cognitive and mobility impairments. Tremors, mainly at rest, slowness of movement (bradykinesia), limb rigidity, and issues with gait and balance are typical motor disorders related to Parkinson’s disease. Additionally, due to progressive muscle atrophy, these issues can lead to falls, which in turn result in further complications and risks to quality of life.
- The Biomaterials, Biomechanics, and Tissue Engineering (BBT) Research Group at the Institute for Health Research and Innovation (IRIS) of the Universitat Politècnica de Catalunya – BarcelonaTech (UPC) is leading DYNAMIC, a project aimed at creating multifunctional, stimuli-responsive biomaterials that not only promote bone regeneration but also intelligently and effectively combat bacterial infections.
- 30/09/2024Project Headerrightno-repeat;left top;;auto20px A team from the Centre for Research in Biomedical Engineering (CREB) of the UPC and Sant Joan de Déu has created a new […]




